
PDF) Validation of the ICP-MS Method for Determination of Trace Elements Provided by ICH Q3D Guideline in Fosamprenavir Calcium

PDF) Validation of the ICP-MS Method for Determination of Trace Elements Provided by ICH Q3D Guideline in Fosamprenavir Calcium
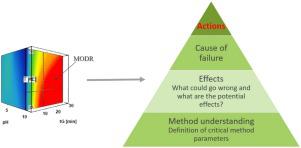
Development of an analytical method for the determination of pimavanserin and its impurities applying analytical quality by design principles as a risk-based strategy,Journal of Pharmaceutical and Biomedical Analysis - X-MOL
















