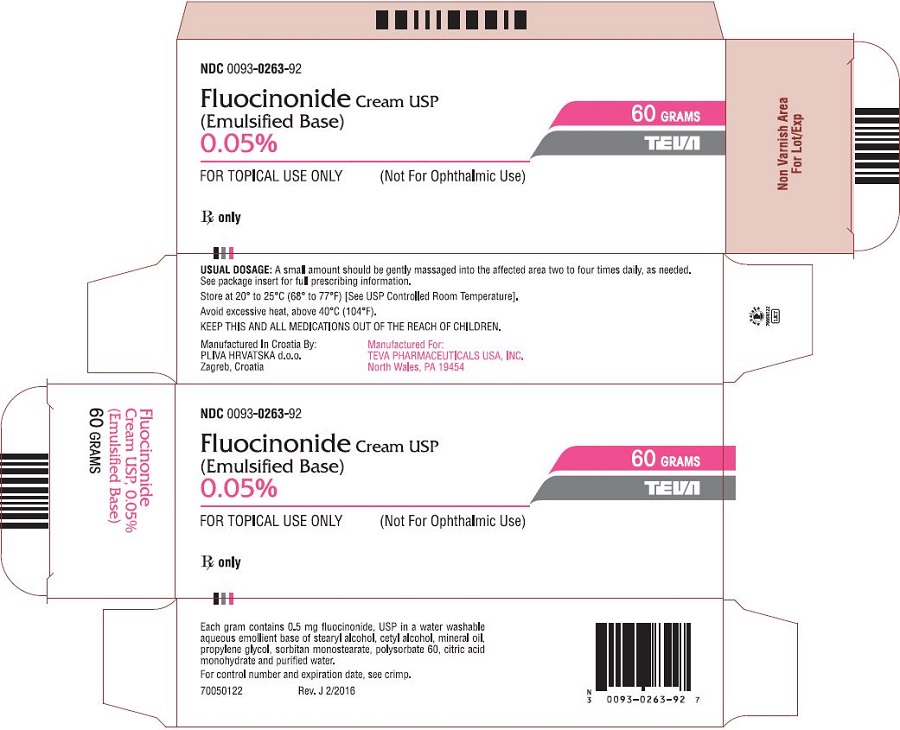
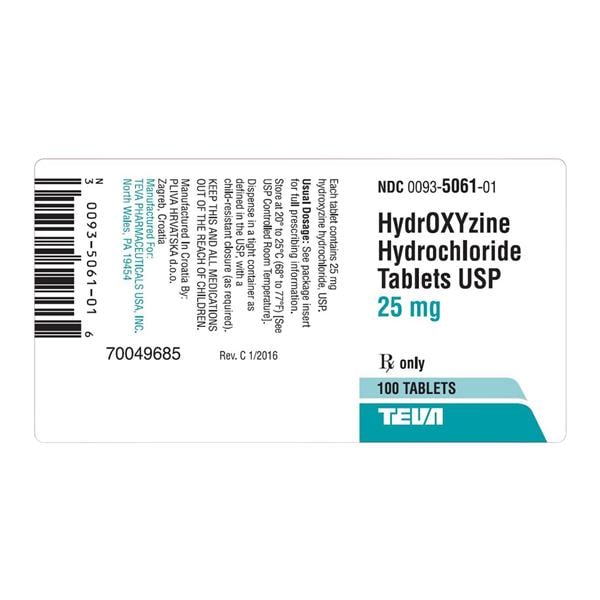
Teva's recall of U.S.-made drugs latest example of contamination fears in generic marketplace; report says Lilly, Pfizer and former Mylan plant in Morgantown have been cited in the past | WV News | wvnews.com

A Teva Pharmaceutical Industries logo is seen on prescription pharmaceutical packaging photographed in a pharmacy Stock Photo - Alamy

Teva Pharmaceutical Industries Limited (TEVA) - Pharmaceuticals & Healthcare - Deals and Alliances Profile | PDF | Initial Public Offering | Pharmaceutical Industry