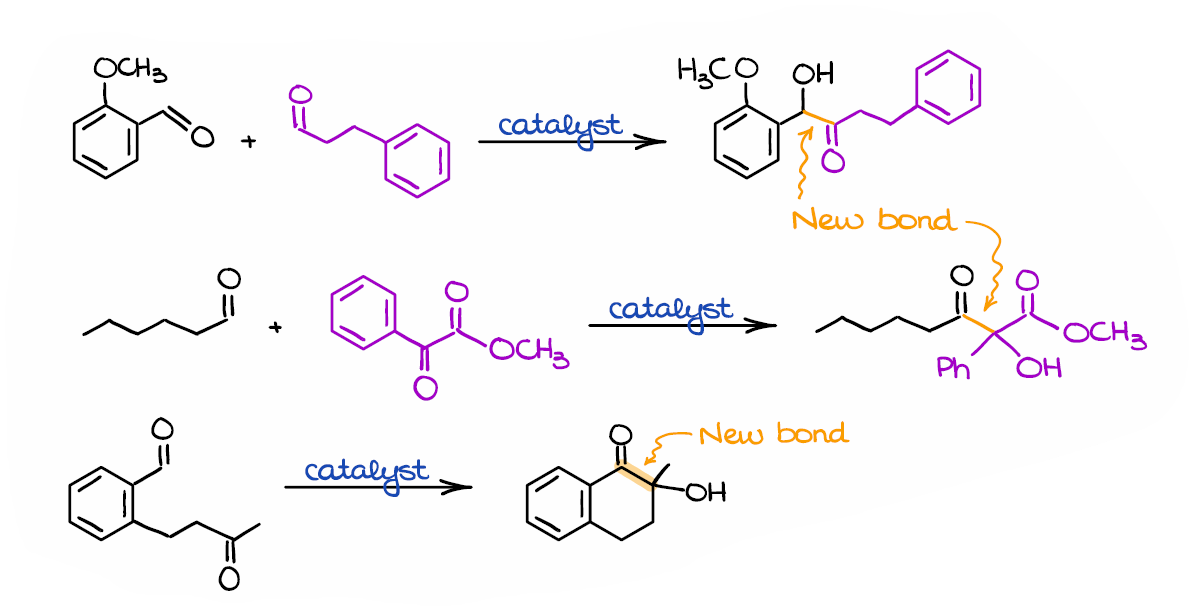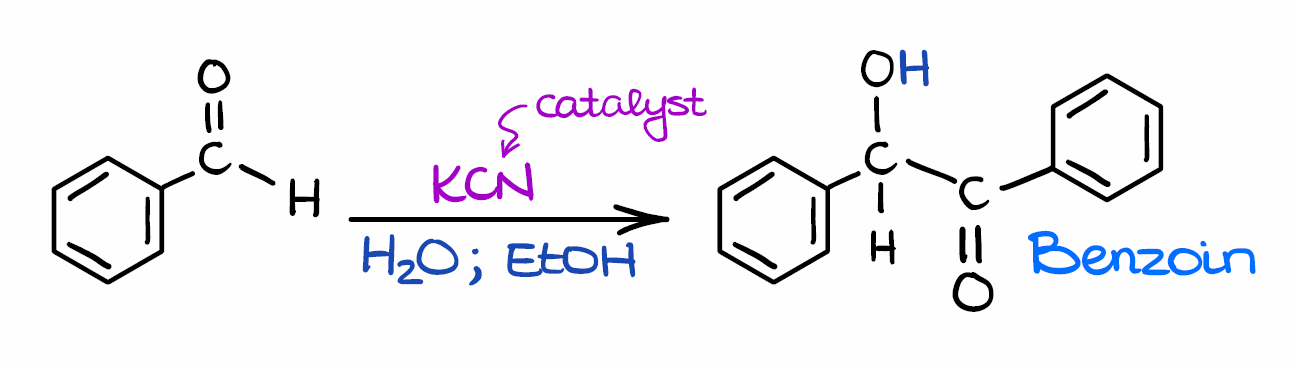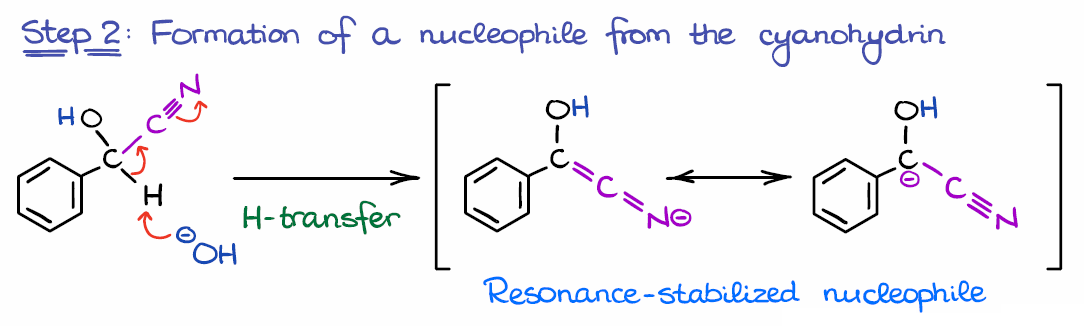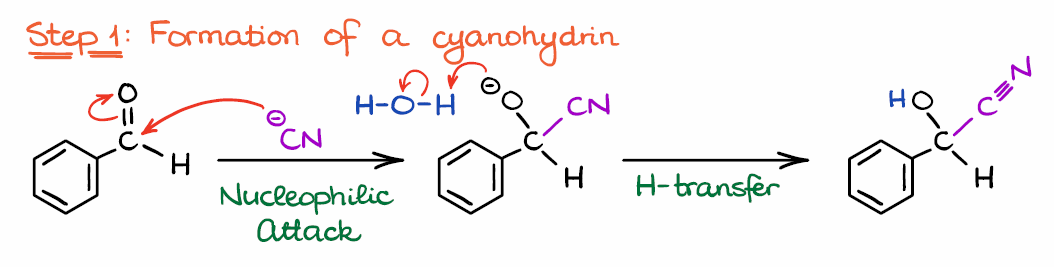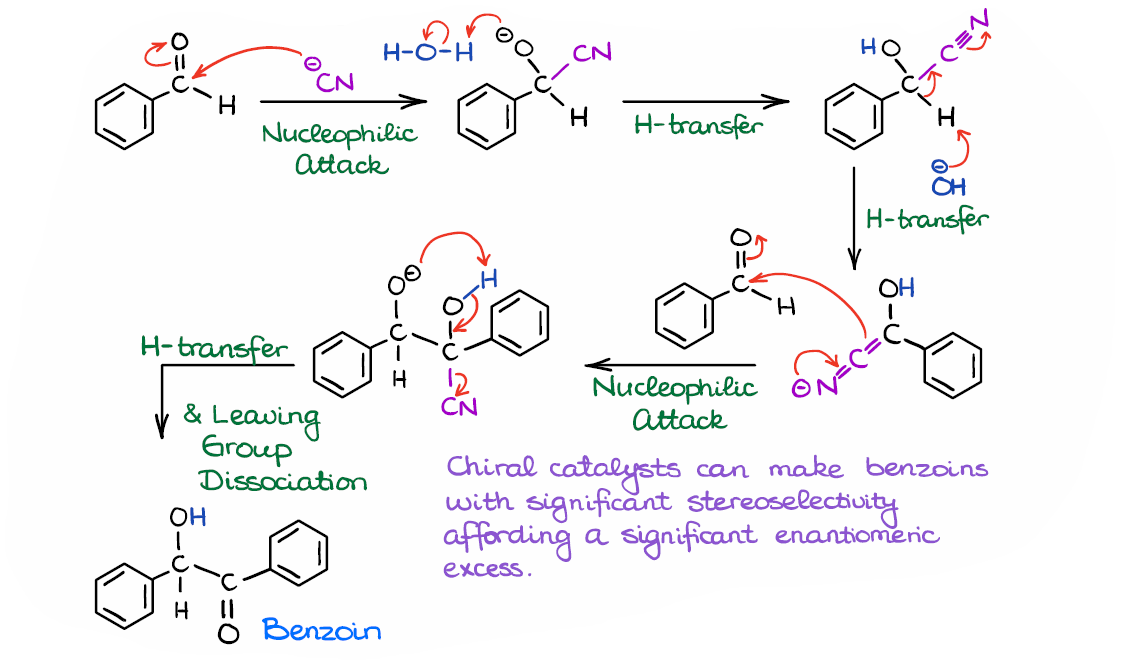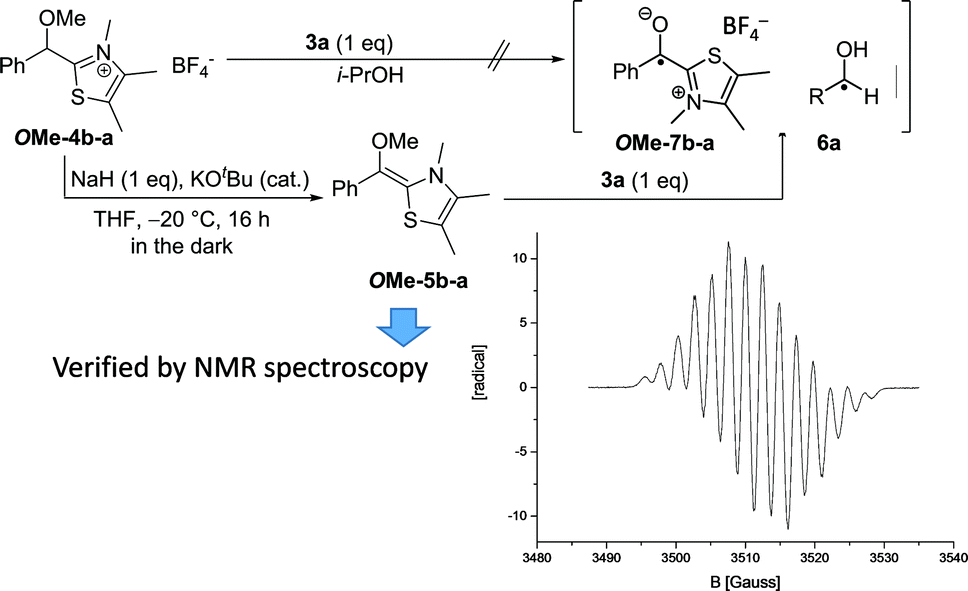
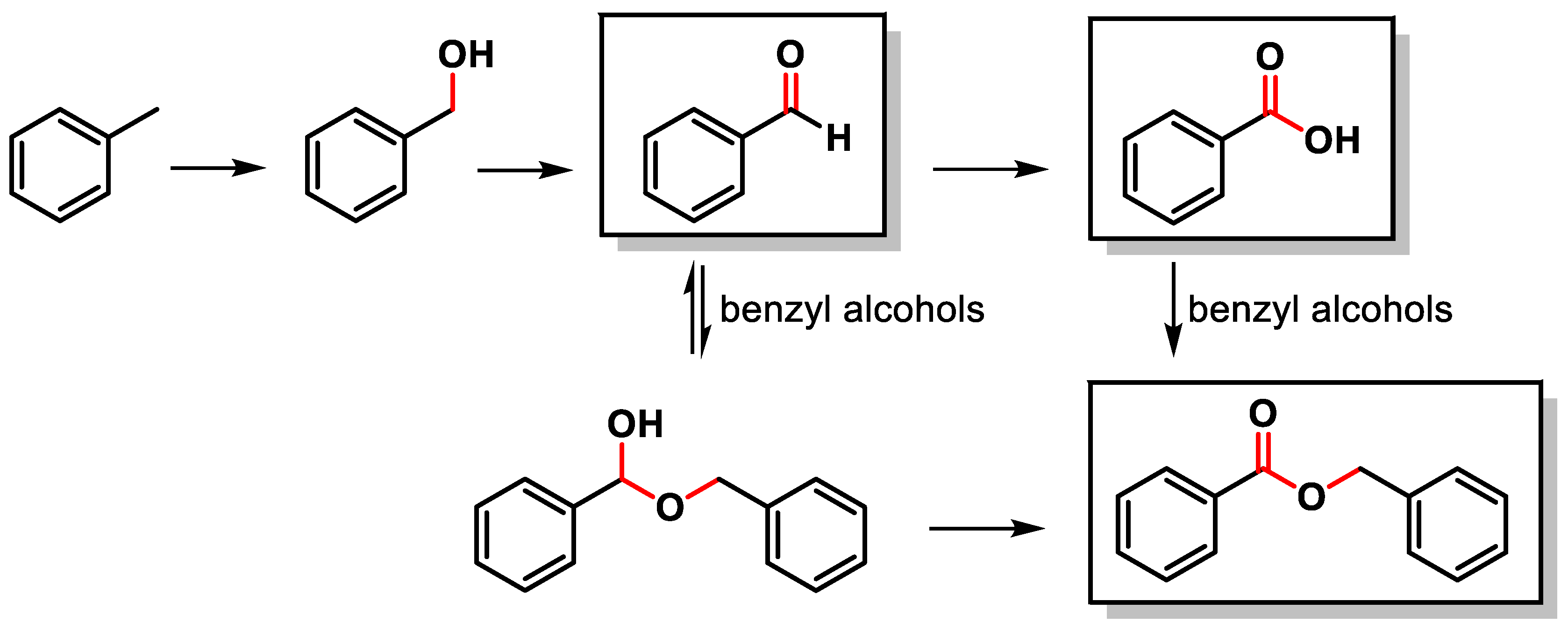
NHC-catalysed benzoin condensation – is it all down to the Breslow intermediate? - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02186C

Catalysis with N‐Heterocyclic Carbenes under Oxidative Conditions - De Sarkar - 2013 - Chemistry – A European Journal - Wiley Online Library

Small Molecule Activation Chemistry of Cu–Fe Heterobimetallic Complexes Toward CS2 and N2O | Inorganic Chemistry
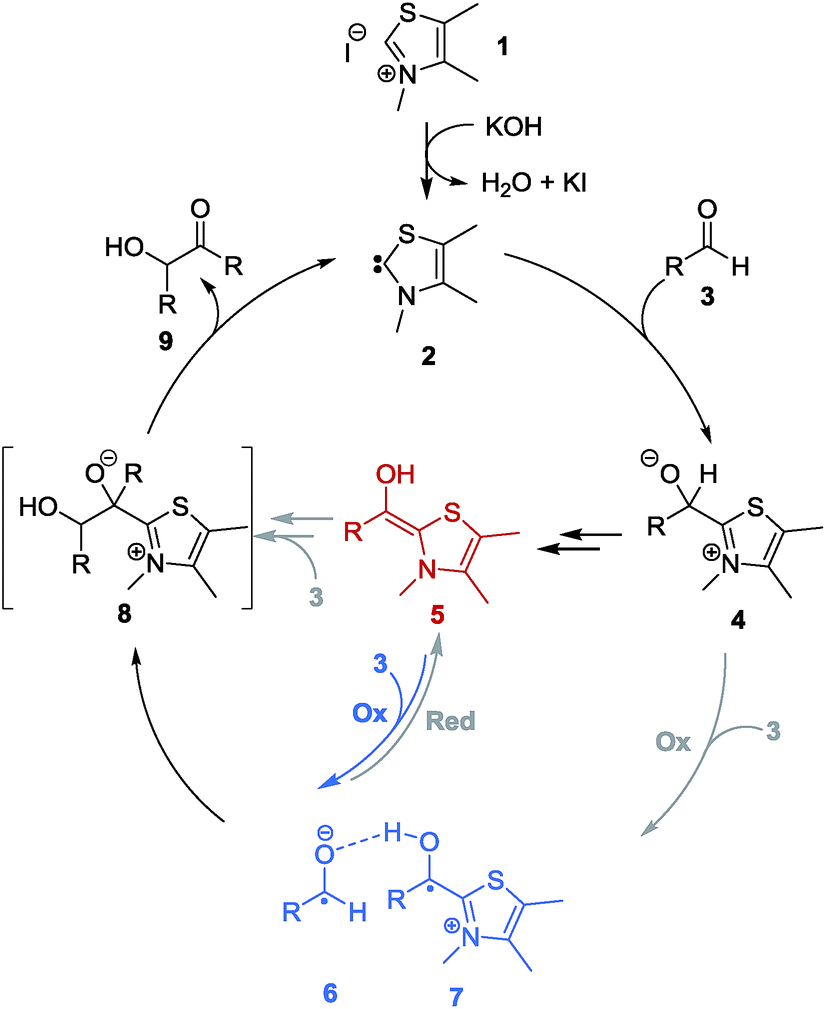
NHC-catalysed benzoin condensation – is it all down to the Breslow intermediate? - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02186C
Catalysts | Free Full-Text | Metal and Metal Oxide Nanoparticles Catalyzed C–H Activation for C–O and C–X (X = Halogen, B, P, S, Se) Bond Formation
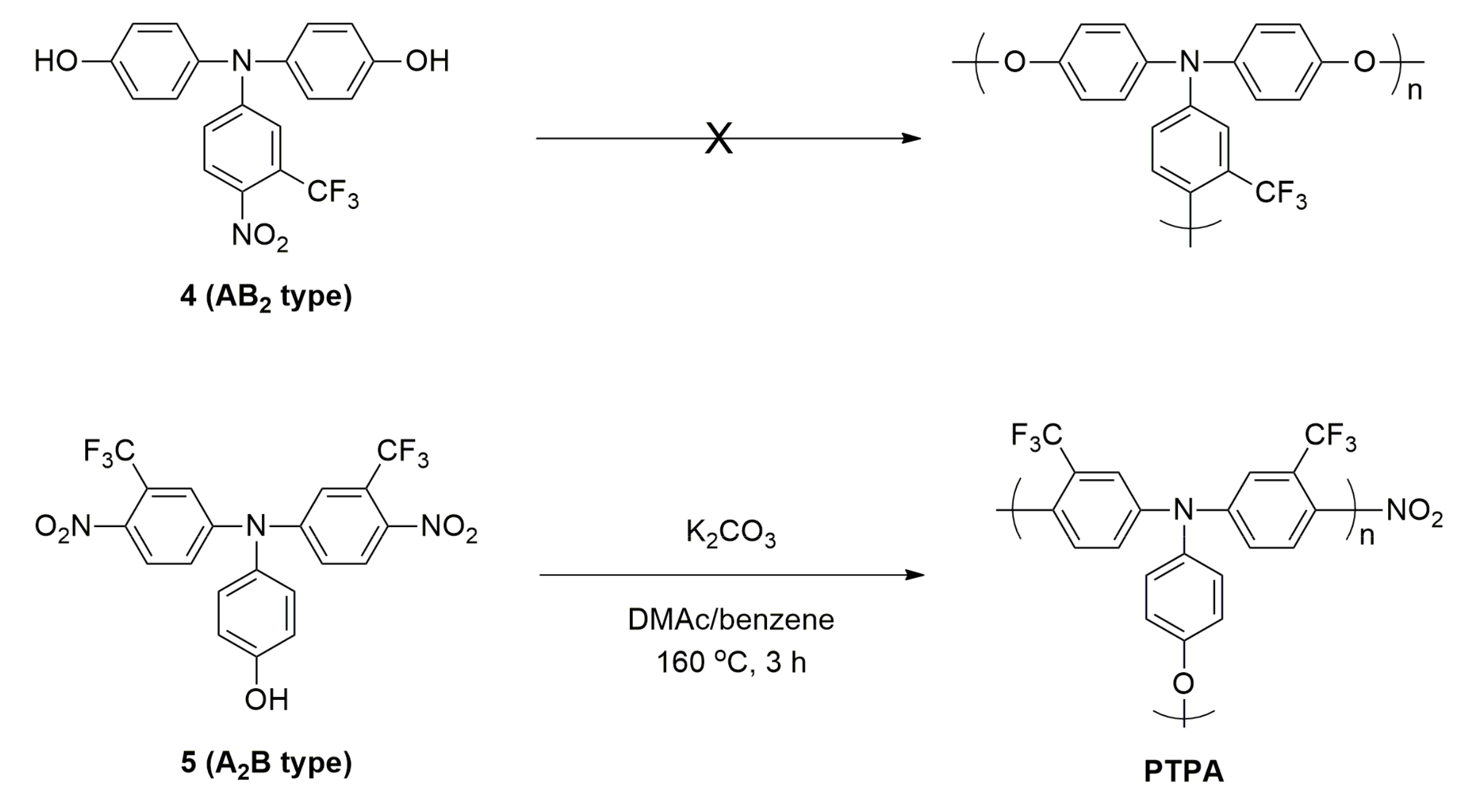
Materials | Free Full-Text | Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes

Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

The Enzymology of Organic Transformations: A Survey of Name Reactions in Biological Systems. - Abstract - Europe PMC

Can the Radical Channel Contribute to the Catalytic Cycle of N-Heterocyclic Carbene in Benzoin Condensation? | The Journal of Organic Chemistry

Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

What Are the Radical Intermediates in Oxidative N-Heterocyclic Carbene Organocatalysis? | Journal of the American Chemical Society

Ruthenium-Catalyzed Intramolecular Hydrocarbamoylation of Allylic Formamides: Convenient Access to Chiral Pyrrolidones | Journal of the American Chemical Society

Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

Can the Radical Channel Contribute to the Catalytic Cycle of N-Heterocyclic Carbene in Benzoin Condensation? | The Journal of Organic Chemistry

Unexpected Direct Synthesis of Tunable Redox-Active Benzil-Linked Polymers via the Benzoin Reaction | ACS Applied Polymer Materials
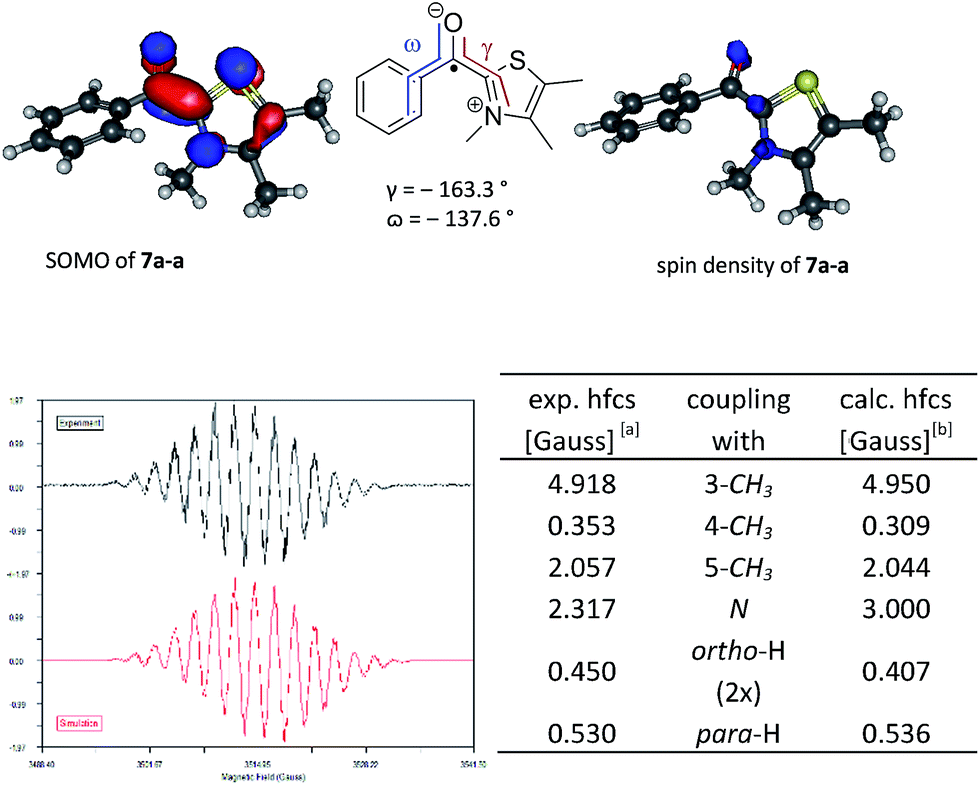
NHC-catalysed benzoin condensation – is it all down to the Breslow intermediate? - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02186C